Mobile clinics are expanding access to care by helping health care organizations deliver services directly to communities.
From medcitynews.comSearch
Pressure, texture, even temperature — transmitted to nerves.
#MedTech
1
1
5
Need a precise low-flow liquid transfer solution? 💧
Meet the 350 Mini Peristaltic Pump:
🔹 5-95ml/min
🔹 100,000+ cycle life
🔹 12V/24V
🔹 REACH/RoHS
Ideal for #MedTech, smart appliances, and labspinmotor.net/350-mini-peris…EtnWn2
#MicroPump #Engineering #PinM5uKFf

1
✨From exposure to capability! ✨
🔸Biomedical engineering students from@VivekanandhaW &@mepcoeng experienced real-world #MedTech systems at@AP_MedTechZone and were introduced to@UBMCinfo.
🔸Join the movementforms.gle/gmzrbxgyoUpYx5…VV
#BiomedicalEngineering #GlobalHealtc2

1
8
革新的デバイス。GlucotrakがQ2にIDE申請予定、2026年後半に米国で埋め込み型血糖モニターの治験開始を目指す。CGM市場の新たな競合として注目される動き。 medtechdive.com/news/glucotrac… #CGM #糖尿病 #MedTech @MedTechDive
Glucotrack plans US trial of implantable glucose monitor this year
Glucotrack plans to file for an investigational device exemption in the second quarter, with hopes of starting a trial in the second half of 2026.
From medtechdive.com 4
242
$XT4 NanoEcho: Revolutionary Cancer Diagnostics #nanoecho #medtech #Annual discdvl.com/compare?q=UEOu…
Disclosure Devil | Analysis
Comparative analysis of financial disclosures by NanoEcho AB
From discdvl.com 7
✨This week, Dr @rathod287, the General Manager for Policy and Strategic Affairs with @InnvolutionH, highlights how #AMTZ is redefining the global #MedTech landscape.
✨"From supporting startups to large-scale manufacturing, #AMTZ is a premier medtech park offering comprehensive to academia, MSMEs, and large companies."
Watch here: youtu.be/RsqdSsjTeSo?si…
#MedTech #AMTZ #HealthcareInnovation #WHIF #HealthTech
1
33
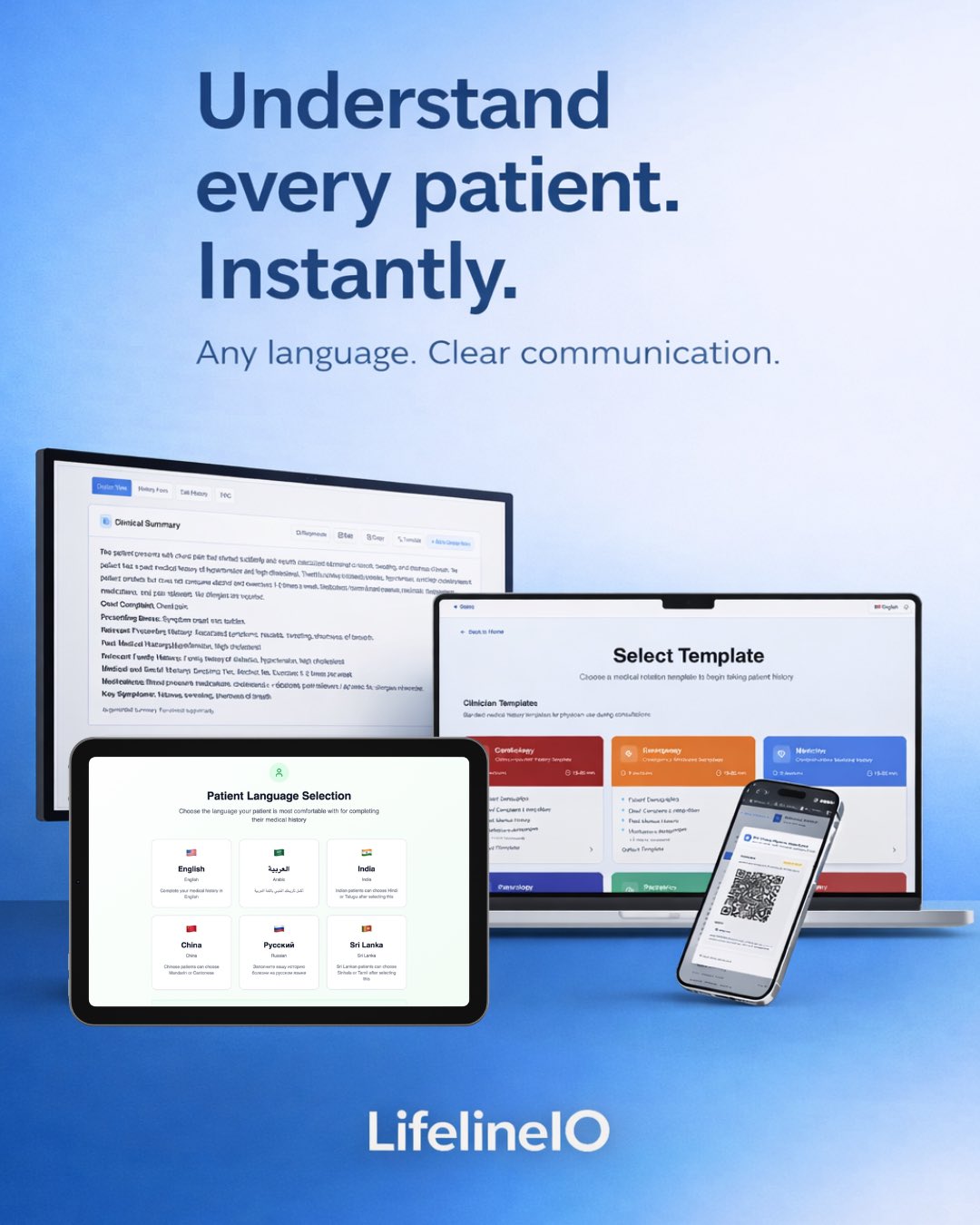
Hot take:
If you don’t understand the patient,
you’re guessing.
Not practicing medicine.
So why is language still treated like a side issue in healthcare?
LifelineIO fixes that.
#AI
#MedTech #DigitalHealth #PatientSafety #AIinHealthcare #HealthInnovation #CAIStartups
4
This isn't just a party trick! Nitinol, the metal in bendable glasses, is revolutionizing medicine. Doctors use Nitinol stents to prop open blocked arteries. #Nitinol #MedTech #Innovation
3
General AI is a risk; Clinical AI is a tool. 🏥 Our custom Micro LLMs are integrated into the clinician's workflow to summarize charts and highlight priorities. It’s about giving doctors back time, not more screens. Precision AI for precision care. #ClinicalAI #MedTech8

1
هل الذكاء الاصطناعي يقدر يكون طبيبك المتنقل؟🤔🩺
قريبا ... بالابحاث و الادلة حنتكلم مع بعض عن الموضوع ده#MedDevice
#MedicalDevice
#MedTech
#Healthit
#mHealth
#HCSM
#Digitalhealth
#Healthcarur

1
20
I’m a urologic oncology fellow @SUO_YUO , not a full-time engineer.
I wanted a faster, cleaner way to log surgical cases, so I built Raivek for iPhone and Apple Watch.
Completely private. No data collected.
Now live on the App Store:
apps.apple.com/us/app/raivek/…
#Raivek #MedTech
2
5
54
4.2K
💡 Healthcare
Mapping cis-regulatory mutations at scale in sorghum enables modulation of gene expression
feeds.nature.com/nbt/rss/current7
#MedTech #HealthTech
3
15
供給網危機。ホルムズ海峡封鎖懸念で原油価格が今月47%急騰、ポリプロピレンも24%上昇。医療機器メーカーの輸送コストとプラスチック材料費が直撃される。地政学リスクがMedTech業界のコスト構造を根底から揺さぶる展開。medtechdive.com/news/manufactu… #MedTech #供給網 #医療機器 @MedTechDive
Manufacturers brace for price increases from Strait of Hormuz closure
Crude oil prices have surged 47% this month and polypropylene jumped 24%, affecting transportation and plastics costs.
From medtechdive.com 2
393
Access Full Report : nextmsc.com/report/physiol…
Download FREE Sample :nextmsc.com/physiological-…
#PhysiologicalSaline #MedTech #HealthcareInvesting #MarketResearch #OutpatientCare #SupplyChain
3
Xcath sees Iris robotic platform transforming #stroke treatment
#medtech #neurology #telesurgery
bioworld.com/articles/72991…
Xcath sees Iris robotic platform transforming stroke treatment
As robotic-assisted surgery platforms increasingly demonstrate that they can perform telesurgery safely, the question now is how soon remote operations will become part of routine clinical care....
From bioworld.com 270
FDA Finalizes Guidance On Including Patient Preference Information In Device Submissions — Click the image below to read more! || #MedTech Insights | Start your free trial today: vist.ly/4wf8i vist.ly/4wf8h
FDA Finalizes Guidance On Including Patient Preference Information In Device Submissions
The US FDA has published final guidance on patient preference information to enhance its evaluation of medical devices. The agency says the guidance strengthens patient engagement in the development...
From insights.citeline.com 25
Are You Addicted to Your Phone? American Phone Usage & Screen Time Statistics - Harmony Healthcare IT
harmonyhit.com/phone-screen-t…
#medtech #healthcare #medicaltechnology #medicine #digitalhealth #telemedicine #wearabletech

4